All published articles of this journal are available on ScienceDirect.
Antiretroviral Drugs as Emerging Environmental Contaminants in South African Water Resources
Abstract
The extensive use of antiretroviral drugs (ARVs) for HIV/AIDS treatment in South Africa has generated a new class of persistent contaminants within the aquatic environment. This review consolidates current evidence on the occurrence, transformation products, ecotoxicological effects, and removal efficiencies of ARVs in South Africa’s water resources. Measured concentrations of compounds such as efavirenz, nevirapine, and lamivudine frequently exceed international ecotoxicological thresholds, reflecting the limitations of conventional wastewater treatment systems designed primarily for nutrient and organic load removal. Because ARVs are only partially metabolized in humans and exhibit high polarity and chemical stability, they persist in effluents, surface waters, and occasionally in drinking water. Chronic exposure studies indicate potential adverse effects on aquatic organisms across trophic levels, including oxidative stress, endocrine disruption, developmental abnormalities, and altered enzymatic activity. The review critically evaluates advanced treatment approaches such as photocatalytic advanced oxidation processes (AOPs), highlighting their efficiencies, scalability, and cost implications for South African contexts. Major research and policy gaps include limited long-term ecotoxicological data, inadequate nationwide monitoring, and the absence of ARV-specific water quality thresholds. The study proposes the integration of ARV monitoring into South Africa’s Green Drop framework, the adoption of visible-light-driven photocatalysis for energy-efficient remediation, and alignment of national water policies with the UN Sustainable Development Goals. By explicitly linking environmental contamination with public health resilience and equity, this review underscores the urgent need for coordinated interventions to ensure that the life-saving benefits of antiretroviral therapy do not compromise aquatic ecosystem integrity.
1. INTRODUCTION
Ecological degradation driven by the continuous release of synthetic organic contaminants, including active pharmaceutical ingredients (APIs), is a growing global concern (Anbarasu and Yuvarajan, 2025; Ashiwaju et al., 2023; Fekadu et al., 2019). Among APIs, antiretroviral drugs (ARVs) are particularly significant due to their widespread and long-term use in the management of human immunodeficiency virus (HIV). South Africa hosts the world’s largest antiretroviral therapy (ART) programme, a cornerstone in transforming HIV/AIDS from a fatal disease into a manageable chronic condition. In 2024, an estimated 40.8 million people were living with HIV globally, with approximately 31.6 million receiving ART (UNAIDS, 2025). South Africa alone accounted for about 8.15 million people living with HIV (12.9% of its total population) (Statistics South Africa, 2025). South Africa is estimated to contribute about 20% (~4 tons per day) of global ARV consumption (Ncube et al., 2018). While ARVs are indispensable for improving survival and quality of life among people living with HIV (PLHIV), their widespread and sustained use has generated an under-recognized environmental concern, the continuous and poorly regulated release of ARVs and their transformation products (TPs) into aquatic ecosystems. ARVs undergo environmental transformations, such as oxidation to carboxylic acids or sulfoxides, forming TPs.
Unlike other pharmaceuticals, ARVs are often excreted partially metabolized (Milburn et al., 2017; Ngumba et al., 2016). They are used in combination therapies and possess physicochemical persistence that renders them resistant to removal in conventional wastewater treatment plants (WWTPs). Fig. (1) illustrates the conceptual pathway of ARVs from human consumption to potential environmental and human health exposure. ARVs enter the environment through partially treated wastewater from WWTPs, discharges from hospitals and pharmaceutical manufacturing, improper disposal of unused or expired medications, and surface runoff, especially in regions lacking adequate sanitation infrastructure (Minguez et al., 2016; Ncube et al., 2018; Peng et al., 2014). Notably, conventional WWTPs are the major source of ARVs into the environment, as they were not designed to remove micropollutants such as ARVs at concentrations in the ng/L–µg/L range (Altaf et al., 2022; Kim and Zoh, 2016). Consequently, their removal is often incomplete and compound-specific. For instance, in conventional wastewater treatment, biological treatment followed by chemical phosphorus removal removed ARVs to varying extents, with abacavir (>99%), stavudine (>78%), and lamivudine (>76%) being largely eliminated, while nevirapine and zidovudine largely bypassed these processes (Prasse et al., 2010). Consequently, aquatic organisms are continually exposed to ARVs at variable concentrations, raising concerns about their potential toxicity in aquatic environments. Detectable concentrations of ARVs and TPs have been reported in influent, effluent, surface waters, sediments, and, in some cases, treated drinking water (Abafe et al., 2018; Nyoni et al., 2024; Schoeman et al., 2017; Wood et al., 2015). A recent government statement further confirmed the presence of ARVs, including nevirapine and efavirenz, in both surface and treated drinking water, particularly downstream of WWTPs (The Water Research Commission (WRC), 2025). This persistent contamination is exacerbated by improper disposal, healthcare system losses, and underperforming wastewater infrastructure, as well as the absence of ARV-specific regulatory guidelines in South Africa (Khan et al., 2020).
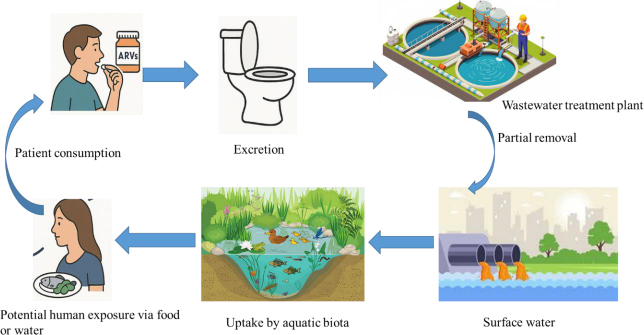
Pathway of ARVs from human use to environmental and human re-entry.
Furthermore, metabolites of nevirapine (12-hydroxy-Nevirapine) and efavirenz (8,14-dihydroxy-Efavirenz) have been detected in South African WWTPs, with higher concentrations in influent than effluent, influenced by season, effluent type, and treatment technology (Mosekiemang et al., 2019). Excreted metabolites can act as secondary pollutants and undergo further modifications, as seen with acyclovir forming carboxy-acyclovir and N-(4-carbamoyl-2-imino-5-oxoimidazolidin) formamido-n-methoxy-acid (COFA) (Schlüter-Vorberg et al., 2015). These TPs resist conventional filtration and have been detected in German rivers, WWTPs, and drinking water (Prasse et al., 2011, 2010). Other ARV TPs, including carboxy-lamivudine (16 ng/L) and carboxy-abacavir (750 ng/L), have been measured in rivers at ng/L levels (Funke et al., 2016). Further, in Germany, carboxy-acyclovir has been detected at concentrations up to 3,200 ng/L in surface water and 40 ng/L in drinking water. This highlights the persistence of ARVs and their TPs and potential environmental exposure as they bypass wastewater treatment. However, limited data on their environmental fate and behavior currently prevent definitive conclusions regarding their long-term impacts and restrict comprehensive ecological risk assessments.
Beyond their occurrence, ARVs exhibit physicochemical properties (e.g., polarity, persistence, and protein binding) and pharmacodynamic characteristics (e.g., antiviral targets that may cross-react with non-target organisms) that can induce chronic, sub-lethal effects across trophic levels. Studies increasingly document the adverse effects of ARVs on aquatic biota, particularly algae, invertebrates, and fish, with evidence of bioaccumulation potential and mixture interactions (Choudhury et al., 2024; Mahaye and Musee, 2022; Nibamureke and Wagenaar, 2021; Souza-Silva et al., 2025b). As ART coverage continues to expand, environmental loading of ARVs and their TPs is expected to persist, highlighting the need for science–policy responses that balance the life-saving benefits of ART with environmental protection.
While global reviews exist on ARV contamination, this is the first comprehensive review that integrates South Africa-specific occurrence data, local ecotoxicological studies, risk quotients, and context-appropriate advanced treatment options, linking these directly to national policy frameworks (e.g., Green Drop system) and SDG alignment. It synthesizes the current state of knowledge on ARVs in South African water resources with four key objectives: (1) to summarize measured environmental concentrations (MECs) of priority ARVs across water matrices; (2) to evaluate ecotoxicological effects and risk assessment (3) to critically appraise removal efficiencies of conventional and advanced treatment technologies such as chlorination, ozonation, advanced oxidation processes (AOPs), activated carbon, membrane bioreactors, nanofiltration/reverse osmosis, and moving bed biofilm reactors and their operational considerations; and (4) to identify regulatory gaps and propose interim thresholds and monitoring priorities aligned with South Africa’s policy landscape (Department of Water and Sanitation, 2023) and international guidance (European Medicines Agency, 2024).
A structured literature search was conducted on the following databases: PubMed, Scopus, Web of Science, Google Scholar, with a focus on South Africa or relevant international context, reporting measured concentrations, toxicity, or treatment, and time frame (primarily 2012–2025. Inclusion criteria prioritized studies reporting robust environmental concentrations, treatment or removal efficiencies, and ecotoxicological outcomes for ARVs and their TPs. By synthesizing occurrence data, ecological impact endpoints, and viable treatment options relevant to resource-limited contexts, this review aims to guide practitioners, regulators, and researchers in prioritizing interventions with maximal ecological and societal benefit. Ultimately, it underscores that closing existing knowledge gaps is essential to maintaining the balance between safeguarding the health of people living with HIV through ART programmes and protecting the integrity of aquatic ecosystems.
1.1. The Issue of Pharmaceutical Pollution
Pharmaceutical contamination of aquatic environments is a growing global concern, but ARVs present distinct challenges that set them apart from other pharmaceutical compounds. In South Africa, home to the world’s largest ARV treatment program, the scale, diversity, and persistence of ARVs in the environment are unmatched. ARVs are typically administered as part of combination therapy regimens, meaning multiple compounds such as efavirenz, lamivudine/emtricitabine, and tenofovir are consumed simultaneously. These drugs often have different physicochemical properties (e.g., solubility, polarity, persistence), which influence their environmental behaviour and toxicity. Moreover, many ARVs are only partially metabolized by the human body and are excreted in active or bioactive forms, including their metabolites (Milburn et al., 2017; Ngumba et al., 2016). For example, dolutegravir, darunavir, and tenofovir are excreted up to 94 and 80% of the parent drugs, respectively, without biotransformation (Kim et al., 2017; Riska et al., 1999). Tipranavir is excreted at 80%, and nevirapine at 2.7% via urine. Assuming a mean of 30% excretion to sewage via urine and faeces, it was estimated that about 159 000 kg of ARVs could reach the aquatic systems of South Africa every year (Swanepoel et al., 2015). This means that ARVs inevitably end up in rivers, lakes, and even drinking water supplies, potentially causing significant environmental harm and contributing to the development of pharmaceutical-resistant microorganisms.
Another concern is the formation of TPs during wastewater treatment, which may possess unknown or increased toxicity. These TPs are rarely monitored, leading to potential underestimation of environmental risks. Research has shown that lamivudine and tenofovir degradation products can inhibit bacterial and algal growth at concentrations as low as 10–50 ng/L (Souza-Silva et al., 2025a). Similarly, preliminary evidence suggests that exposure of Raphidocelis subcapitata to acyclovir and its TPs, carboxy-acyclovir and COFA (secondary TP), for 72 hours showed that COFA exerted the strongest inhibitory effect, significantly reducing algal yield and growth rate, with EC10 and EC50 values of 4.12 mg/L and 18.15 mg/L, respectively. Reported laboratory studies within South Africa reported that in Daphnia magna exposed for 21 days, carboxy-acyclovir significantly reduced reproduction and population growth by 39.9% and 22.4%, respectively, indicating higher toxicity than the parent compound. In contrast, COFA TP exhibited no significant toxicity in Daphnia. Further, no acute fish toxicity for acyclovir and both TPs up to ~100 mg/L (Schlüter-Vorberg et al., 2015). Overall, these findings indicate that while COFA poses the greatest risk to algal growth, carboxy-acyclovir exerts more pronounced chronic toxicity in Daphnia, whereas acyclovir and its TPs show minimal acute toxicity to fish, highlighting species-specific sensitivities and differential ecological risks among the compounds.
Although numerous degradation processes for ARVs have been explored (Madikizela, 2025). In the last decade, research on the identification of antiretroviral drugs in water has intensified, especially in developing countries. Concentrations reaching 33 µg/L for efavirenz have been reported in wastewater effluent, which is released into the surface water. Some antiretroviral drugs have been found to possess toxic effects on aquatic organisms and plants. This threatens the sustainability of high-quality vegetables as the water scarcity necessitates the practise of crop irrigation with used or treated water, which is often contaminated with pharmaceuticals. This article reviewed the progress made on the removal of antiretroviral drugs in contaminated water, identified challenges and gaps, while outlining the possible future research directions. It was observed that various degradation processes for antiretroviral drugs have been explored, with limited information on the characterization of the degradation by-products and their toxic effects. The adsorption process is also dominating for removal of antiretroviral drugs in water, while wetland removal and plant uptake are the least explored. An adsorption capacity as high as 833 mg/g has been reported for lopinavir using biochar modified by incorporating layered double hydroxide. Research on the removal of antiretroviral drugs in water is expected to continue with the inclusion of studies focusing on metabolites of these chemicals. Madikizela (Madikizela, 2025), emphasizes that a major challenge remains: the identification and toxicity assessment of their TPs. This difficulty arises from low environmental concentrations, complex matrices, and the need for advanced analytical expertise, typically requiring GC–MS or LC–MS techniques and a strong understanding of organic transformation pathways, highlighting the need for further research on ARV degradation and by-product toxicity. Unlike broader categories of pharmaceuticals, the environmental risks of ARVs are amplified by their chronic, low-level presence in regions with high HIV prevalence and their lack of removal during conventional treatment processes.
1.2. The Impact on Water Resources
The introduction of ARVs into environmental matrices presents considerable risk to both public health and the environment. Even though concentrations detected in aquatic systems are frequently minimal, prolonged exposure, even at trace levels, may lead to detrimental health outcomes. A critical yet often neglected issue is the potential for the development of drug resistance, akin to resistance patterns observed with antibiotics, if ARVs are not effectively eradicated from the environment. Persistent environmental exposure could facilitate the selection of resistant strains of HIV or related viruses, thereby undermining decades of progress in HIV clinical management. Such resistance would pose severe challenges for South Africa, a nation combating the world’s largest HIV epidemic and one that has made extensive financial and infrastructural investments in treatment programs to achieve the World Health Organization’s treatment for all and 95-95-95 objectives. The escalation of ARV usage in alignment with these targets is anticipated to increase drug loads in the environment, thereby heightening this risk. The emergence of resistant strains would undermine the efficacy of first-line and second-line regimes, necessitating more costly and complex treatment regimens, thereby imposing additional burdens on the already overextended healthcare system.
From an ecological perspective, the presence of ARVs in the environment poses significant disruptions to aquatic ecosystems. Empirical research has demonstrated that ARVs interfere with the hormonal systems of fish, consequently altering their reproductive cycles and behaviours (Nibamureke et al., 2019b; Nibamureke and Wagenaar, 2021). These disruptions extend beyond fish populations, cascading through the food web, resulting in bioaccumulation in wildlife and posing risks to human populations dependent on aquatic organisms for nutrition. In the long term, the intersection of ecological and health ramifications of ARV contamination may plausibly lead to biodiversity loss, further destabilization of aquatic ecosystems already burdened by climate change and pollution, and reduced resilience of these systems. Ultimately, without the implementation of effective strategies for the monitoring and removal of ARVs from the environment, the sustainability of both public health initiatives and ecosystem integrity remains at risk.
1.3. Occurrence of ARVs in Aquatic Ecosystems
Recent South African studies have documented the occurrence and persistence of ARVs in surface waters and wastewater effluents, as shown in Fig. (2) and Table S1-S5. For example, (Schoeman et al., 2017) reported that effluent discharged from a wastewater treatment plant in Southern Gauteng contained concentrations ranging between 5 500 to 14 000 ng/L for efavirenz and 92 and 743 ng/L nevirapine, indicating that conventional treatment processes are inadequate for removing these compounds. Similarly, (Nibamureke and Barnhoorn, 2025) screened surface waters in the Vhembe District, Limpopo
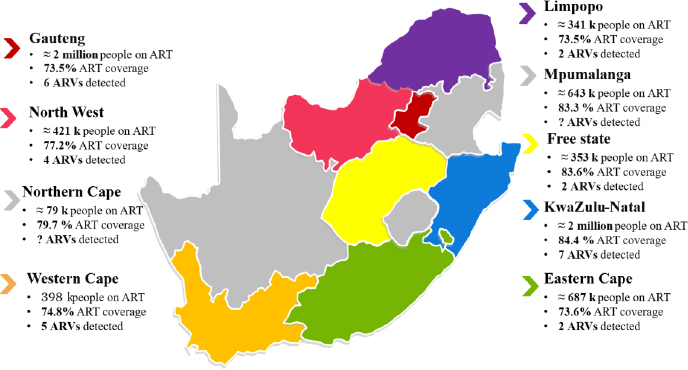
Number of people on ART, ART coverage, and number of ARVs detected in the environment in South Africa.
Province, and found nevirapine and lopinavir concentrations reaching up to 166 and 42 ng/L, respectively. These levels exceed reported ecotoxicological thresholds for aquatic organisms such as fish and invertebrates, underscoring the need for site-specific risk assessments. (Swanepoel et al., 2015) evaluated the prevalence of ARVs in wastewater, surface water, groundwater, and drinking water across different regions of South Africa. The study reported the presence of 12 ARVs in different environmental compartments. The highest concentrations were reported in Kwa-Zulu Natal Province, attributed to the highest HIV statistics. This further highlights the widespread and persistent nature of ARV contamination.
The Water Research Commission report (Nyoni et al., 2024) evaluated the removal of contaminants of emerging concern in drinking water and wastewater treatment systems. They reported that, while conventional processes such as coagulation, sedimentation, and chlorination achieved only partial reductions of pharmaceuticals, ARVs were resistant to removal. By contrast, advanced treatment processes such as ozonation, activated carbon adsorption, and membrane technologies demonstrated substantially higher removal efficiencies, although cost and scalability remain major challenges. Complementing these findings, a recent study by the North-West University (NWU) (North West University, 2025) quantified ARV residues in South African water resources and reported concentrations in some cases exceeding acceptable thresholds, posing potential long-term health risks. The study further highlighted the inadequacy of existing wastewater treatment processes and recommended ARV-specific regulatory guidelines, improved treatment infrastructure, and multidisciplinary research to address ecological and human health impacts. Together, these reports underscore both the technological and regulatory gaps that must be bridged to protect water quality and public health in South Africa. The research further highlighted alarming effects on aquatic ecosystems and wastewater management systems. For instance, freshwater snails exposed to ARVs exhibited altered embryonic development (North West University, 2025). Further, concentrations of ARVs ranging from 670 to 34 000 ng/L (influents) and 540 to 34 000 ng/L (effluents) were determined in WWTPs in South Africa compared with Europe. In Europe, reported concentrations ranged from less than the limit of detection (LOD) to 32 ng/L (influents) and less than LOD to 22 ng/L (effluents) (Adeola and Forbes, 2022). These findings emphasize the urgent need for tailored environmental monitoring programs and stronger policy interventions to address the environmental presence of ARVs in South African water systems.
These compounds can affect aquatic organisms at various trophic levels, from primary producers like algae, and consumers like daphnids, to higher consumers such as fish (Choudhury et al., 2024; Ngwenya et al., 2025; Souza-Silva et al., 2025b). However, little is known about their effects on non-target aquatic organisms to draw meaningful conclusions. Furthermore, reported concentration ranges should be interpreted with caution due to differences in analytical methodologies, sample preparation techniques, and limits of detection/quantification across variant studies.
Effects on primary producers (e.g., algae) are particularly concerning, as these organisms form the base of aquatic food webs. Exposure to ARVs has been shown to inhibit photosynthesis and reduce algal growth, which may have cascading effects on the food chain (Gomes et al., 2022; Souza-Silva et al., 2025a). Daphids are also important species in freshwater food chains as primary consumers. Their exposure to ARVs has been reported to cause immobilization, mortality, effects on reproduction, biochemical markers, induce oxidative stress, etc (Mahaye and Musee, 2022; Souza-Silva et al., 2025a). Additionally, secondary consumers, such as fish, which have great economic and dietary importance and subsequently serve as a major doorway to human exposure, have exhibited endocrine disruption, altered reproductive success, and behavioral changes due to ARV exposure (Choudhury et al., 2024; Nibamureke and Wagenaar, 2021). While much of the research focuses on the acute toxicity of ARVs, long-term effects, particularly the chronic exposure of aquatic organisms to sub-lethal concentrations, remain poorly understood. Importantly, current studies contribute to understanding the ecotoxicological impacts of ARVs and highlight the importance of long-term assessments to adequately estimate environmental risks of ARVs.
1.4. Ecotoxicity of ARVs in Aquatic Biota
Over the years, considerable attention has been devoted to the investigation of the toxicity of ARVs in mammalian systems (Carr and Cooper, 2000; Hawkins, 2010). It is only in recent times that research has started to elucidate the ecotoxicity of these pharmaceuticals, a factor that contributes to the limited availability of ecotoxicity data for ARVs (Olabode and Somerset, 2019) The toxicity of ARVs toward aquatic organisms, such as fish, algae, daphnia, and bacteria, is classified among the top ten most toxic categories of pollutants (Sanderson et al., 2004). However, only a limited number of studies have reported toxicity data such as acute and chronic endpoints or sub-lethal effects across different levels of biological organization Table 1. Moreover, existing findings are often inconclusive, largely because most experiments were conducted in synthetic media rather than environmentally relevant conditions. This paucity of hazard characterization underscores an urgent need for systematic ecotoxicological assessments of ARVs in non-target species. Although extensive data exist on the environmental occurrence of ARVs across various matrices (Table S1-S5), there remains no clear risk assessment framework addressing their potential impact on ecosystem integrity or human health through exposure pathways such as food or drinking water. For instance, ARVs have already been detected in drinking water supplies (Wood et al., 2015), yet no regulatory threshold values have been established to guide safety evaluations.
| ARV | Organism | Exposure (h) | Exposure concentrations (mg/L) | E(L)C50/NOEC (mg/L) | Findings | Reference |
|---|---|---|---|---|---|---|
| Abacavir | Daphnia magna | 48 | 0.000003826 | > 100 | Non-toxic, EC50 >100 mg/L. | (Minguez et al., 2016) |
| Abacavir | Pseudokirchneriella subcapitata | 72 | 0.000003826 | 57.32 | Harmful, 10 mg/L<EC50<100 mg/L. | (Minguez et al., 2016) |
| Acyclovir | Daphnia magna | 48 | 0.000128223 | 64.12 | Harmful, 10 mg/L<EC50<100 mg/L. | (Minguez et al., 2016) |
| Acyclovir | Pseudokirchneriella subcapitata | 72 | 0.000128223 | >100 | Non-toxic, EC50 >100 mg/L. | (Minguez et al., 2016) |
| Acyclovir | Raphidocelis subcapitata | 96 | 1.25 - 20 | 3.612 | Significantly inhibited algal growth at 2.5 to 20 mg/L. | (Almeida et al., 2021) |
| Acyclovir | Ceriodaphnia dubia | 192 | 1.25 - 20 | 3.062 | Significant fertility decreases in females exposed to concentrations of 2.5 to 20 mg/L. | (Almeida et al., 2021) |
| Acyclovir | Raphidocelis subcapitata | 72 | 200 | Not determined | No toxicity was observed up to 100 mg/L | (Schlüter-Vorberg et al., 2015) |
| Acyclovir | Daphnia magna | 504 | 200 | Not determined | No mortality or reduction in reproduction or population growth was observed. | (Schlüter-Vorberg et al., 2015) |
| Acyclovir | Danio rerio | 96 | 200 | Not determined | No acute fish toxicity up to ∼100 mg/L. | (Schlüter-Vorberg et al., 2015) |
| Carboxy-acyclovir (TP) | Raphidocelis subcapitata | 72 | 200 | Not determined | Non-toxic to algae | (Schlüter-Vorberg et al., 2015) |
| Carboxy-acyclovir (TP) | Daphnia magna | 504 | 200 | Not determined | Significantly reduced reproduction by 40% at 102 mg/L. | (Schlüter-Vorberg et al., 2015) |
| Carboxy-acyclovir (TP) | Danio rerio | 96 | 200 | Not determined | No acute fish toxicity up to ∼100 mg/L. | (Schlüter-Vorberg et al., 2015) |
| COFA (2nd TP) | Raphidocelis subcapitata | 72 | 200 | 18.15 | COFA inhibited the growth of green algae with an EC10 of 14.1 mg /L. | (Schlüter-Vorberg et al., 2015) |
| COFA (2nd TP) | Daphnia magna | 504 | 200 | Not determined | COFA was not toxic to Daphnia. | (Schlüter-Vorberg et al., 2015) |
| COFA (2nd TP) | Danio rerio | 96 | 200 | Not determined | No acute fish toxicity up to ∼100 mg/L. | (Schlüter-Vorberg et al., 2015) |
| Atazanavir | Echinometra lucunter | 1 | 3.12 - 100 | 73.04 | Significant reduction in fertilization is only at high concentrations. | (Cid et al., 2021) |
| Atazanavir | Echinometra lucunter | 42 | 0.195 - 3.12 | 0.78 | Delayed or abnormal larval development at ≥0.5 mg/L. | (Cid et al., 2021) |
| Dolutegravir | Microcystis novacekii | 96 | 0.1 | 1.7 | No significant cell growth inhibition, but it reduced the metabolic activity of cyanobacteria. | (Souza-Silva et al., 2025a) |
| Dolutegravir | Microcystis novacekii | 336 | 0.1 | 0.4 | Exposure time directly influenced inhibition of cell growth, leading to a significant toxicity increase, M. novacekii was unable to recover from intoxication by DTG. | (Souza-Silva et al., 2025a) |
| Efavirenz | Daphnia magna | 48 | 0.625 - 1 | > 1 | No mortality, but significant changes in CAT and GST activities, and MDA levels | (Mahaye and Musee, 2022) |
| Efavirenz | Echinometra lucunter | 1 | 3.12 - 100 | 11.46 | Decreased fertilization success at lower concentrations than others. | (Cid et al., 2021) |
| Efavirenz | Echinometra lucunter | 42 | 0.195 - 3.12 | 0.195 | Malformed larvae and developmental delay | (Cid et al., 2021) |
| Efavirenz | Oreochromis mossambicus | 96 | 10.3 - 20.6 | 0.0000000103 | Liver damage and overall decline in fish health | (Robson et al., 2017) |
| Efavirenz | Raphidocelis subcapitata | 96 | 0.008 - 0.25 | 0.034 | Toxicity decreased in the order of EFV >3TC > ACV > ZDV. Significantly inhibited algal growth from 0.016 - 0.25 mg/L. | (Almeida et al., 2021) |
| Efavirenz | Ceriodaphnia dubia | 192 | 0.031 - 0.5 | 0.026 | Significant decrease in fertility at concentrations of 0.031 to 0.25 mg/L. | (Almeida et al., 2021) |
| Efavirenz | Synechococcus elongatus | 96 | 0.0003 - 0.1 | 0.04012 | EFV significantly decreased growth rate at concentrations of ≥0.3 μg/L. | (Gomes et al., 2022) |
| Efavirenz | Chlorococcum infusionum | 96 | 0.0003 - 0.1 | 0.01107 | Reduced growth rate, impaired photosynthesis, altered respiration, and disrupted oxidative metabolism. | (Gomes et al., 2022) |
| Lamivudine | Raphidocelis subcapitata | 96 | 1.25 - 20 | 3.013 | Significantly inhibited algal growth at 2.5 to 20 mg/L. | (Almeida et al., 2021) |
| Lamivudine | Ceriodaphnia dubia | 192 | 0.625 - 10 | 1.345 | Significant reduction in neonates at concentrations from 1.25 to 10 mg/L. | (Almeida et al., 2021) |
| Lamivudine | Daphnia magna | 24 | 0.01 - 0.1 | 0.0341 | 10 and 85% mortality at 0.01 and 0.1 mg/L, respectively. | (Omotola et al., 2023) |
| Lamivudine | Daphnia magna | 48 | 0.01 - 0.1 | 0.0123 | 45 and 100% mortality at 0.01 and 0.1 mg/L, respectively. | (Omotola et al., 2023) |
| Lamivudine | Microcystis novacekii | 96 | 100 | 184.8 | Very low toxicity: even 100 mg/L did not significantly alter metabolic activity. | (Souza-Silva et al., 2025a) |
| Lamivudine | Microcystis novacekii | 336 | 100 | > 400 | No inhibition was observed. | (Souza-Silva et al., 2025a) |
| Lamivudine | Synechococcus elongatus | 96 | 0.0003 - 0.1 | 570.3 | No effects on any parameters that were evaluated. | (Gomes et al., 2022) |
| Lamivudine | Chlorococcum infusionum | 96 | 0.0003 - 0.1 | 571.1 | Like S. elongatus, none of the LMV tested (up to 0.1 mg/L) showed effects on growth rate and biochemical endpoints. | (Gomes et al., 2022) |
| Lopinavir | Danio rerio | 96 | 0.1 - 40 | 6.42 | Conjugation with PEG showed improved solubility and activity with reduced toxicity compared to pure LPV. | (Aremu et al., 2020) |
| Lopinavir | Danio rerio | 96 | 10 | 0.099-0.744 | Under photo-induced processes, lopinavir may cause pericardial edema and shortening of the tail, affecting its behavior. | (Czech et al., 2022) |
| Nevirapine | Echinometra lucunter | 1 | 3.12 - 100 | 84.61 | Minimal effects on fertilization at environmentally relevant levels. | (Cid et al., 2021) |
| Nevirapine | Echinometra lucunter | 42 | 0.195 - 3.12 | 0.39 | Strong developmental inhibition even at low concentrations. | (Cid et al., 2021) |
| Nevirapine | Oreochromis mossambicus | 720 | 0.00148 | 0.00000148 | No adverse effects on larvae hatching, survival, and behavior | (Nibamureke et al., 2019) |
| Nevirapine | Oreochromis mossambicus | 1440 | 0.00148 | Not reported | Insignificant lower mean growth rate compared to controls | (Nibamureke et al., 2019b) |
| Nevirapine | Oreochromis mossambicus | 720 | 0.00148 - 0.00374 | Not reported | The mean Hb concentrations in exposed fish were slightly lower than in the controls | (Nibamureke et al., 2019c) |
| Stavudine | Daphnia magna | 48 | Toxicity predicted using (Q)SAR model | 980 | Non-toxic, EC50 >100 mg/L. | (Sanderson and Thomsen, 2009) |
| Tenofovir | Microcystis novacekii | 96 | 40 - 300 | 161.01 | Resistant to Tenofovir, tolerating concentrations > 100 mg/L | (Silva et al., 2019) |
| Tenofovir | Artemia salina | 24 | 30 - 180 | 111.82 | Induced immobility | (Silva et al., 2019) |
| Tenofovir | Alivibrio fischeri | 0.25 | 4.28 - 70.4 | 14.8 | Inhibition of luminescence | (Silva et al., 2019) |
| Tenofovir | Daphnia magna | 48 | 0.0625 - 1 | > 1 | Concentration- and time-dependent immobilization > 0.25 mg/L | (Mahaye and Musee, 2022) |
| Tenofovir | Ceriodaphnia dubia | 24 | 6.25 - 100 | 2.83 | Tenofovir induced ~29.98% mortality at 100 mg/L indicating nontoxicity | (Nugnes et al., 2024) |
| Tenofovir | Brachionus calyciflorus | 24 | 6.25 - 100 | 100 | Induced 50% mortality | (Nugnes et al., 2024) |
| Tenofovir | Raphidocelis subcapitata | 72 | 0.02441 | 0.04812 | Harmful during chronic exposure | (Nugnes et al., 2024) |
| Tenofovir | Brachionus calyciflorus | 48 | 0.009 - 10 | 0.00108 | Exhibited the highest sensitivity starting from very low concentrations | (Nugnes et al., 2024) |
| Tenofovir | Ceriodaphnia dubia | 168 | 6.25 - 100 | 0.0574 | Ritonavir was the most toxic, followed by tenofovir, then ribavirin | (Nugnes et al., 2024) |
| Tenofovir | Biomphalaria glabrata | 72 | 3 - 300 | Not determined | No mortality was observed. | (Souza-Silva et al., 2023) |
| Tenofovir | Biomphalaria glabrata | 504 | 3 - 300 | 2.7 | An increase in hemocytes' metabolic and phagocytic activity caused behavioral changes. | (Souza-Silva et al., 2023) |
| Tenofovir | Microcystis novacekii | 96 | 10 | 130.7 | No effect on cellular growth | (Souza-Silva et al., 2025a) |
| Tenofovir | Microcystis novacekii | 336 | 10 | 147.0 | 12% reduction in its metabolic activity at 10 mg/L, indicating that the cell can suffer effects even when there is no reduction in its cellular growth | (Souza-Silva et al., 2025a) |
| Tenofovir | Synechococcus elongatus | 96 | 0.0003 - 0.1 | 594.4 | No effects on any of the parameters evaluated. | (Gomes et al., 2022) |
| Tenofovir | Chlorococcum infusionum | 96 | 0.0003 - 0.1 | 671.1 | Like S. elongatus, none of the TNF tested (up to 0.1 mg/L) showed effects on growth rate and biochemical endpoints. | (Gomes et al., 2022) |
| Ribavirin | Brachionus calyciflorus | 24 | 0.009 - 10 | 3.23 | Induced 50% mortality | (Nugnes et al., 2024) |
| Ribavirin | Ceriodaphnia dubia | 24 | 1.56 - 200 | 71.08 | Only ribavirin achieved a full LC50 | (Nugnes et al., 2024) |
| Ritonavir | Danio rerio | 96 | 10 | 0.061-0.601 | The number of dead embryos and noted malformations was strictly linked to increased concentration. | (Czech et al., 2022) |
| Ritonavir | Brachionus calyciflorus | 24 | 0.3 - 100 | 2 | Ritonavir, with LC50s at tens of mg/L, was considered harmful to B. calyciflorus | (Nugnes et al., 2024) |
| Ritonavir | Ceriodaphnia dubia | 24 | 0.156 - 2 | 11.35 | The highest tested concentration (2 mg/L) produced ~38.33% mortality | (Nugnes et al., 2024) |
| Ritonavir | Raphidocelis subcapitata | 72 | 0.02441 | 0.02293 | Harmful to algae under chronic exposure | (Nugnes et al., 2024) |
| Ritonavir | Brachionus calyciflorus | 48 | 7.8 x 10-8 - 0.03 | 0.0002 | Caused a higher chronic effect, determining 50% of inhibition of reproduction | (Nugnes et al., 2024) |
| Ritonavir | Ceriodaphnia dubia | 168 | 0.00867 - 0.01 | 0.1432 | Ritonavir was identified as the most toxic drug | (Nugnes et al., 2024) |
| Zidovudine | Daphnia magna | 48 | Toxicity predicted using (Q)SAR model | < 100 | Harmful, EC50 <100 mg/L. | (Sanderson and Thomsen, 2009) |
| Zidovudine | Ceriodaphnia dubia | 192 | 1.25 - 20 | 5.671 | Synergistic effects at concentrations of 1.25 and 2.5 mg/L were observed. | (Almeida et al., 2021) |
| Zidovudine | Raphidocelis subcapitata | 96 | 1.25 - 20 | 5.442 | Significant increase in the microalga at 1.25 mg/L, indicating a hormesis effect. | (Almeida et al., 2021) |
Ecotoxicity data in Table 1 reveal highly variable responses of aquatic organisms to ARVs. Toxicity ranged from non-toxic to highly toxic effects and was compound-, organism-, and exposure duration-dependent. For example, acyclovir and abacavir generally showed low to moderate toxicity, with EC50 values often >50 mg/L and in several cases >100 mg/L. However, acyclovir transformation products (e.g., carboxy-ACV) exhibited increased toxicity at chronic exposures (e.g., reduced Daphnia reproduction). In contrast, efavirenz, ritonavir, and lopinavir demonstrated higher toxicity, with sub-lethal effects (e.g., oxidative stress, developmental abnormalities) and EC50/LC50 values in the low mg/L to µg/L range. Dolutegravir showed time-dependent toxicity, increasing with prolonged exposure, particularly in cyanobacteria. Lamivudine and tenofovir generally displayed low toxicity, with several studies reporting no significant effects at environmentally relevant concentrations. However, acute effects were observed in Daphnia magna and Brachionus calyciflorus at prolonged exposures. Nevirapine showed limited acute toxicity but caused developmental inhibition in echinoderm larvae. Transformation products posed differential risks, with some, such as carboxy-ACV, being more toxic than their parent compounds. Overall, toxicity trends in Table 1 suggest species-specific sensitivities, with microalgae, rotifers, and early developmental life stages often being the most vulnerable, particularly to efavirenz, ritonavir, and dolutegravir.
Almeida et al., (Almeida et al., 2021) found that efavirenz exhibited an EC50 and NOEC of 0.026 mg/L and 0.016 mg/L, respectively, when exposed to Ceriodaphnia dubia over eight days, illustrating that efavirenz is extremely toxic to C. dubia. Robson et al., (Robson et al., 2017) discovered that an exposure of 0.0103 µg/L of efavirenz resulted in severe hepatic damage and concomitantly reduced long-chain triglyceride (LCT) levels in Oreochromis mossambicus after 96 hours of exposure, an effect mirroring effects observed in humans (Blas-García et al., 2010). Similarly, chronic toxicity evaluations over 28 days by Pitso (Pitso, 2020) identified marked renal and hepatic toxicity in O. mossambicus. Omotola et al., (Omotola et al., 2021) illustrated that lamivudine is highly toxic to Daphnia magna, with a 100% mortality rate observed at 0.1 mg/L following 48 hours of exposure (Omotola et al., 2021). Additionally, chronic toxicity spanning eight days, conducted using lamivudine on C. dubia yielded an EC50 of 1.345 mg/L and a NOEC of 0.625 mg/L, highlighting the toxicity of the drug to Daphnia (Almeida et al., 2021). Minguez et al., (Minguez et al., 2016) exposed abacavir to D. magna and Pseudokirchneriella subcapitata for 48 and 72 hours, respectively, finding EC50s of >100 mg/L and 57.32 mg/L, thereby indicating non-harmful and harmful toxicity, respectively. Conversely, zidovudine, was also categorized as toxic to daphnia (C. dubia) with an EC50 of 5.671 mg/L (Almeida et al., 2021).
In addition to the dearth of individual drug toxicity data, the combined effects of parent drugs and their TPs may exhibit even higher toxicity towards aquatic life (Gosset et al., 2021; Kumar et al., 2021). However, such investigations are significantly lacking, despite the extensive use of these drugs in sub-Saharan Africa. This underscores the imperative for systematic investigations into the toxicity of ARVs across different levels of biological organization. Considering the high costs and time-intensive nature of quantifying the environmental risks associated with pollutants, such as the multitude of ARVs, modelling approaches are essential to estimate, for instance, their predicted environmental concentrations (PECs) to quantify exposure levels (Keller, 2006; Keller et al., 2014). Furthermore, models can be employed to estimate the aquatic toxicity of most ARVs utilizing the Ecological Structure Activity Relationships (ECOSAR) software, thereby facilitating the calculation of predicted no-effect concentration (PNEC) values necessary for risk assessment.
1.5. Ecological Risk Assessment
The frequent detection of ARVs such as EFV, NVP, 3TC, TDF, and AZT in WWTP influent and effluent in South Africa, as well as in the associated receiving surface waters, represents an increasing ecotoxicological concern. To evaluate the plausible risk posed by these pharmaceuticals in environmental matrices, the risk quotient method (Equation 1) was employed. The ECOSAR and NORMAN models were utilized to estimate the predicted no-effect concentrations (PNECs), as shown in Fig. (3), while measured environmental concentrations (MECs) reported in the literature were used. The risk categorization was conducted using the framework proposed by (Hernando et al., 2006). According to this framework, an RQ < 0.01 indicates no ecological risk, ARV drugs with 0.01 ≤ RQ < 0.1 present low risk, those with 0.1 ≤ RQ < 1 present minimal risk, and pollutants with an RQ ≥ 1 present a high ecological risk.
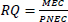
For instance, the MEC of EFV, reaching up to 140.4 µg/L in influents and 37.3 µg/L in effluents, as well as 3TC concentrations of up to 20.9 µg/L, significantly surpass the PNECs for aquatic organisms, notably algae, daphnia, and fish. This potential exceedance under high risk condition suggests the potential for both acute and chronic ecological risks (Abafe et al., 2018; Wood et al., 2015). The persistence of these compounds through conventional treatment processes, along with their presence in surface waters, underscores the inefficacy of removal methods and the potential for bioaccumulation and trophic transfer within aquatic ecosystems.
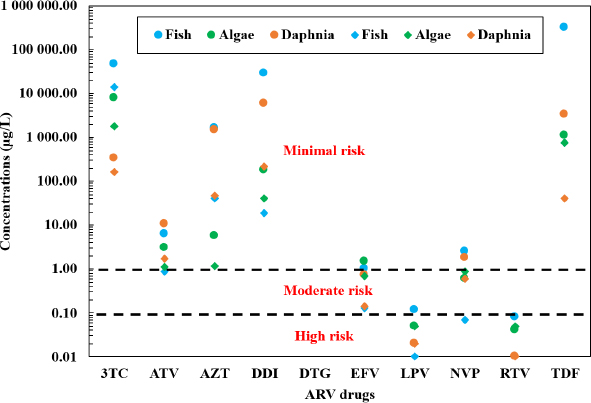
Acute (circles) and chronic (diamonds) Predicted No-Effect Concentrations of ARVs (Ngwenya and Musee, 2023).
Notably, EFV emerges as the compound with the highest environmental risk among the ARVs detected in South African water matrices. Utilizing the surface-water concentration of 38.45 µg/L and a conservative PNEC of 0.20 µg/L (NORMAN, 2025; US EPA, 2015), the RQ of 192.3 indicates a significant ecological risk (Almeida et al., 2021). Comparable observations have been documented globally, where EFV's low biodegradability and pronounced hydrophobicity contribute to its persistence and toxicity in aquatic environments (Ngwenya and Musee, 2023). Chronic exposure investigations further corroborate EFV's capability to induce oxidative stress, reproductive impairments, and developmental toxicity in Daphnia magna and fish species (Mahaye and Musee, 2022). These findings underscore that even at ecologically relevant concentrations, EFV poses a considerable threat to aquatic biota.
Similar to EFV, NVP, and LPV also present moderate to high ecological risks, regardless of low effluent concentrations recorded up to 1.9 μg/L and 3.8 μg/L, respectively. These compounds manifest chronic toxicity to Daphnia magna and fish, even at sub-microgram levels (PNECs < 0.1 μg/L), suggesting that their persistence in aquatic environments could adversely affect reproductive and growth functions in aquatic organisms (Souza-Silva et al., 2025a). Other ARVs, including TDF and 3TC, exhibited lower numerical RQs but remain a concern due to their persistence and chronic effects. For NVP and TDF, RQs of 0.065 and 2.8 × 10−4 were derived using surface-water concentrations of 2.8 µg/L and 0.25 µg/L with PNECs of 43 µg/L and 900 µg/L, respectively, indicating a low acute risk. Nonetheless, chronic toxicity has been documented in Daphnia magna and fish at sub-microgram levels, indicating potential long-term ecological damage (Souza-Silva et al., 2025a). Similarly, 3TC exhibited a low calculated RQ of 0.034, though literature suggests that laboratory-derived PNECs may be substantially lower than regulatory values, implying a potential underestimation of risk (K’oreje et al., 2020; Ngwenya and Musee, 2023).
In addition to direct toxicological effects, the persistent environmental presence of ARVs introduces a supplementary, indirect risk by potentially fostering the development of antiviral drug resistance among environmental microorganisms. Analogous to the effects observed with antibiotics, chronic low-level exposure to ARVs can exert selective pressure on microbial communities, promoting the horizontal gene transfer of resistance determinants to pathogenic strains (Apetroaei et al., 2024). This phenomenon could compromise the efficacy of HIV treatment initiatives, endangering the substantial national investments directed toward HIV management and precipitating novel public health challenges through the establishment of environmental reservoirs of resistance.
In summary, the findings suggest that while certain ARVs, such as 3TC and TDF, exhibit low ecological risk under current conditions, compounds like EFV and LPV remain of significant concern due to their persistence, potential for bioaccumulation, and toxicity even at trace concentrations. The observed elevated RQs underscore the necessity for ongoing environmental monitoring, more stringent regulatory controls, and enhanced wastewater treatment technologies capable of effectively removing ARVs. Moreover, the reliance on ECOSAR predictions, limited chronic toxicity data for South African species, and the need for refining local PNECs and integrating both chronic and mixture toxicity assessments are essential for a more precise evaluation of ecological risk in South African aquatic systems.
1.6. Remediation of Antiretroviral Drugs in Wastewater
The prevalence of ARV drug compounds in South African wastewater underscores the limitations of conventional wastewater treatment methodologies, predominantly based on biological and sedimentation processes. Specific compounds such as EFV, NVP, and lamivudine (3TC) persist post-treatment and are commonly detected in effluent and receiving water bodies at concentrations that pose significant ecological hazards (Ncube et al., 2025). The inherent chemical stability, low biodegradability, and polar nature of these compounds impede their microbial degradation and adsorption, necessitating the implementation of advanced remediation strategies to alleviate both ecological and public health risks.
| ARV | Photocatalyst | Catalyst dosage (g/L) | Irradiation | Removal efficiency (%) | Reference |
|---|---|---|---|---|---|
| 3TC | P25 | 1 | 365 UV | >95 | (An et al., 2011) |
| ABC | GO-TiO2 | 0.1 | 500 W m−2 | 99 | (Evgenidou et al., 2023) |
| ACV | TNPs-MWCNTs | 0,02 | 125 W Hg lamp | 98.6 | (Chen et al., 2014) |
| ACV | g-CN/TiO2 | 0,03 | 300 W Xe lamp | 100 | (Li et al., 2016) |
| ACV | Ag2MoO4/g-C3N4 | 0,25 | 300 W Xe lamp | 100 | (Wu et al., 2018) |
| ACV | Bi4VO8Cl | 1 | 300 W Xe lamp | 100 | (Hu et al., 2014) |
| ACV | TNPs-MWCNTs | 0.4 | 365 | 98 | (Chen et al., 2014) |
| ACV | g-CN/TiO2 | 0.3 | >420 | 100 | (Li et al., 2016) |
| ACV | Ag2MoO4/g-C3N4 | 0.25 | >420 | 100 | (Wu et al., 2018) |
| ACV | BiVO8Cl | 0.05 | 200 – 700 | 100 | (Hu et al., 2014) |
| AZT | 5%BZONPs | 0,1 | 8 W UV lamp | 100 | (Bhamare and Kulkarni, 2019) |
| AZT | CuSm0.06Fe1.94O4@g-C3N4 | 0,12 | Visible | 71.5 | (Masunga et al., 2022) |
| AZT | CuO@Ag@Bi2S3 | 0,02 | 200 UV-A | 87.4 | (Ayodhya, 2022) |
| AZT | CuSm0.06Fe1.94O4@g-C3N4 | 1.2 | >420 | 72 | (Masunga et al., 2022) |
| AZT | CuO@Ag@Bi2S3 | 0.02 | 200 UV-A | 87 | (Ayodhya, 2022) |
| EFV | Ag-AgBr-LDH | 2 | 84 | (Tabana et al., 2023) | |
| LPV | Ammonium molybdate (WU and WW photocatalysts) | 0.4 | 500 – 550 | 95 | (Hojamberdiev et al., 2022) |
| NVP | FL-BP@Nb2O5 | 0.1 | >420 | 68 | (Bhembe et al., 2020) |
| NVP | Ag-AgBr-LDH | 2 | 100 | (Tabana et al., 2023) | |
| RBV | Bi4VO8Cl | 0.05 | 200-780 | 100 | (Hu et al., 2014) |
| RTV | (WU and WW photocatalysts) | 0,4 | 500-550 | 95 | (Hojamberdiev et al., 2022) |
| STV | CuO@Ag@Bi2S3 | 0,02 | 200 UV-A | 92.1 | (Ayodhya, 2022) |
Advanced Oxidation Processes (AOPs), as depicted in Table 2, including photocatalysis and UV/H2O2 treatment, have demonstrated superior efficacy in the degradation of ARVs (Ngwenya et al., 2025; Ruziwa et al., 2023; Zhang et al., 2023). Photocatalytic systems utilizing P25 titanium dioxide (TiO2) for 3TC and graphene oxide-titanium dioxide (GO-TiO2) for abacavir (ABC) have achieved removal efficiencies exceeding 95% under moderate ultraviolet (UV) irradiation conditions (An et al., 2011; Evgenidou et al., 2023). Similarly, compounds such as acyclovir (ACV), AZT, and protease inhibitors, including LPV and ritonavir (RTV), can be effectively degraded using nanocomposites such as TiO2 nanoparticles-multiwalled carbon nanotubes (TNPs-MWCNTs), graphite carbon nitride/titanium dioxide (g-CN/TiO2), bismuth vanadate chloride (Bi4VO8Cl), and silver-silver bromide layered double hydroxide (Ag-AgBr-LDH) under UV or visible light, achieving removal efficiencies exceeding 98% (Cheng et al., 2014; Hu et al., 2014; Li et al., 2016; Tabana et al., 2023).
Previous work has proven that these processes facilitate non-selective oxidation, ensuring near-total mineralization without the formation of persistent metabolites (Dagnew et al., 2025; Yang et al., 2021). AOPs have shown superiority to conventional water and wastewater treatment processes due to the production of reactive radicals capable of degrading chemically stable ARVs, which conventional biological processes fail to efficiently eliminate (Aziz et al., 2025; Xiangyu et al., 2025). Furthermore, the modular design of photocatalytic reactors allows for seamless integration into existing WWTPs with minimal infrastructural modification, such as the retrofitting of UV lamps or immobilized catalysts within secondary treatment stages. This integration is cost-effective and scalable for deployment within existing South African facilities where comprehensive upgrades may be economically restrictive.
The application of photocatalysts activated by visible light and low-power UV systems enhances their suitability within the context of South Africa. Compounds such as CuO@Ag@Bi2S6 and CuSm0.06Fe1.94O4@g-C3N4 demonstrate effective degradation of ARV agents such as AZT, STV, and LPV at reduced catalyst concentrations under moderate irradiation conditions (Ayodhya, 2022; Hojamberdiev et al., 2022; Masunga et al., 2022). These systems effectively decrease operational expenses and energy consumption while maintaining high efficiencies in ARV removal, thereby supporting decentralized or resource-constrained treatment facilities and minimizing the release of residual catalysts. While advanced treatment technologies show promising removal efficiencies in the cited studies, most data originate from laboratory or pilot-scale experiments conducted under controlled conditions, mostly outside South Africa. Direct transferability to full-scale South African WWTPs (with variable influent composition, infrastructure, and operational constraints) should therefore be interpreted with caution.
1.7. Gaps in Research and Policy
Consistent with prior reports (Department of Water and Sanitation, 2023; Nyoni et al., 2024), and as highlighted in the occurrence data, risk assessment, and the North West University (North West University, 2025), the absence of regulatory thresholds for ARVs emphasized the need for long-term ecological and human health impact studies. To guide future action, Table 3 summarizes research priorities crucial for informing risk assessments and policy development in South Africa.
| Category | Gap Identified | Recommendation |
|---|---|---|
| Ecotoxicological Data | Limited long-term, sub-lethal exposure studies across multiple trophic levels | Long-term ecotoxicological studies examine chronic, sub-lethal effects of ARVs on diverse aquatic species. |
| Biodegradation | Poor understanding of ARV degradation pathways and environmental fate | Investigation into the degradation pathways, transformation products, and persistence of ARVs in various environmental compartments (e.g., sediments, groundwater). |
| Mixture Toxicity | Few studies on the combined effects of ARVs with other pollutants | Impact assessments of ARV mixtures and interactions with other environmental contaminants. |
| Monitoring Capacity | Inadequate inclusion of ARVs in national water quality monitoring programs | Expansion of national monitoring programs to routinely detect ARVs in surface and drinking water. |
| Regulatory Framework | Absence of discharge thresholds or ARV-specific environmental regulations | Development of ARV-specific water quality standards, tailored to South Africa’s environmental and public health context. |
1.8. Challenges in Addressing the Issue
There are several factors involved in managing pharmaceutical pollution in water resources. Wastewater treatment plants in South Africa, especially in rural areas, are not designed to remove pharmaceutical residues. Most wastewater treatment processes focus on biological and chemical pollutants. Furthermore, monitoring and detecting ARV contamination in water resources is a difficult task. Standard water testing methods may not be sufficient to identify specific pharmaceutical compounds present. The lack of widespread research on the impact of ARVs in South Africa makes it difficult to fully understand the scope of the problem. There is also limited data on concentration levels of ARVs in water systems, making it challenging to assess risks to public health and the environment. The lack of public awareness regarding the presence of ARVs in water resources is another challenge. South Africa has made significant progress in addressing HIV/AIDS through widespread access to ARVs. However, the public is not aware of the environmental implications of their use. Without greater public education on how pharmaceuticals affect water systems, efforts to mitigate pollution may be insufficient.
1.9. Societal, Ecological, and Health Implications
The persistence of ARVs in South Africa’s water resources has far-reaching consequences that extend beyond ecology into public health and social equity. Ecologically, ARVs disrupt aquatic food webs, reduce biodiversity, and alter ecosystem services such as nutrient cycling and water purification. Bioaccumulation in aquatic organisms poses additional risks to wildlife and to humans who consume fish or other aquatic species.
From a health perspective, chronic low-level exposure to ARVs through contaminated drinking water remains a concern, particularly for vulnerable groups such as children, the elderly, and immunocompromised individuals. Prolonged exposure may also foster the emergence of drug-resistant pathogens, potentially undermining the gains of South Africa’s HIV/AIDS treatment programme.
Socially and economically, unmanaged ARV pollution may exacerbate inequities, as disadvantaged communities dependent on untreated or poorly treated water sources face disproportionate risks. Rising water treatment costs and impacts on livelihoods reliant on aquatic ecosystems further illustrate the societal burden of pharmaceutical pollution.
Addressing ARV contamination is therefore not only an environmental and public health priority but also a development challenge. It directly aligns with the United Nations Sustainable Development Goals (SDGs 3, 6, and 14), underscoring the urgent need for integrated solutions that combine technological innovation, stronger policy frameworks, and community engagement. By recognizing the interconnected ecological, health, and social dimensions of this crisis, South Africa can advance sustainable water management while safeguarding public health and social equity.
1.10. Future Directions and Recommendations
1.10.1. Research Priorities
Future research should prioritize comprehensive monitoring of ARVs across surface water, groundwater, sediments, and drinking water sources. Emphasis is needed on long-term ecological and human health studies, including bioaccumulation, chronic low-dose exposure, and the potential for fostering drug-resistant pathogens. Developing ARV-specific water quality thresholds and toxicity benchmarks is essential for integrating pharmaceuticals into existing national water quality frameworks. Further studies should also investigate the cost-effectiveness and scalability of advanced treatment technologies such as photocatalysis, ozonation, membrane bioreactors, nanofiltration, and moving bed biofilm reactors within the South African context. Multidisciplinary collaborations bridging water science, public health, and social sciences will be critical to fully assess the societal implications of ARV contamination.
1.10.2. Technological Recommendations
Addressing the issue of ARV contamination in South Africa's aquatic systems necessitates the implementation of a comprehensive, multi-dimensional approach, which includes substantial investment in wastewater treatment infrastructure. Conventional wastewater treatment facilities are generally inadequate in eliminating ARVs from effluents. However, the integration of advanced treatment technologies, which have demonstrated high removal efficiencies (>90–98%) for several photocatalysts at laboratory and pilot scale, may enhance the shortcomings of conventional treatment methods. These advanced technologies hold significant promise, attributable to their superior degradation efficiencies. However, full-scale implementation in resource-constrained South African WWTPs would plausibly require further evaluation of catalyst stability, energy requirements, operational costs, and integration feasibility.
1.10.2.1. Photocatalyst
Presents a sustainable and versatile method for the mineralization of persistent ARV compounds into innocuous by-products, thereby preventing secondary environmental contamination (Ncube et al., 2025; Ngwenya et al., 2025; Tabana et al., 2023). In wastewater treatment facilities that do not currently employ UV-based disinfection systems, the incorporation of photocatalysts activated by visible light, such as modified TiO2 or g-C3N4 composites, can effectively utilize solar energy to facilitate the degradation process, thereby reducing both operational costs and energy consumption. In contrast, for facilities already utilizing UV disinfection methods, the integration of UV-responsive catalysts like TiO2 or ZnO can be efficiently integrated within the existing infrastructure, optimizing the use of available UV radiation to improve contaminant removal efficacy. This strategic deployment of photocatalysis, customized to align with the specific configurations of existing treatment plants, offers a cost-effective, energy-efficient, and scalable approach to mitigating the pollution of water resources in South Africa by ARVs.
1.10.2.2. Ozonation
Already implemented in several treatment facilities for disinfection purposes, presents a technically feasible and cost-effective strategy for enhancing the removal efficiency of ARV compounds. Ozone, a potent oxidizing agent, can degrade numerous pharmaceuticals through electrophilic and radical mechanisms. Its incorporation as a tertiary treatment step can significantly diminish micropollutant concentrations with minimal infrastructural alterations (Beek, 2021; Funke et al., 2021). For facilities currently utilizing ozone, optimizing the ozone dosage and contact duration, or upgrading to ozone-based advanced oxidation processes (e.g., O3/H2O2 or O3/UV), can improve the degradation of persistent ARVs (Luu and Lee, 2014). To ensure comprehensive detoxification, ozonation should be succeeded by biologically activated carbon filtration to eliminate transformation products (Lim et al., 2022). Routine monitoring of parent compounds, transformation products, and by-products is crucial for safe operation and ecological protection. When properly optimized, ozonation represents a viable and scalable solution for mitigating ARV contamination in South Africa’s aquatic systems. In a recent study by (Zheng et al., 2024), ozonation successfully degraded oseltamivir phosphate (OP) from variant water matrices, including secondary municipal wastewater effluent. Removing over 90% of OP from a buffer solution using 10 µM of ozone in 30 seconds.
1.10.2.3. Nanofiltration
Nanofiltration (NF) and reverse osmosis (RO) have been recognized as promising technologies for the rejection of pharmaceuticals(Patel et al., 2022; Taheran et al., 2016). (Dolar et al., 2023) showed that RO membranes are the most efficient in removal and show the highest removal of > 90–95% from synthetic waters. In the case of NF membranes, moderate to high removal of pharmaceuticals was reported. The main removal mechanisms in RO/NF include size exclusion, the Donnan effect (electrostatic exclusion), and hydrophobic interactions. In the case of RO, the main removal mechanism is size exclusion since the molecular weight cut-off (MWCO) of RO membranes is 100 Da, while the molecular weight of most pharmaceuticals is larger, i.e., 150–500 g/mol.
1.10.2.4. Membrane Bioreactors (MBRs)
A membrane bioreactor is a combination of a membrane process, like ultra-filtration or nano-filtration, with an activated sludge process. MBRs were reported to remove 100% of acetaminophen within 24 h. When equipped with reverse osmosis and nano-filtration membrane, it removed around 20 and 13 contaminants respectively with > 90% removal efficiency. In addition, increasing some factors, such as sludge retention time, has shown significant removal efficiency while enhancing the performance of the bioreactor. The advantage of MBR over conventional treatment techniques is that it has greater removal efficiency, a small footprint, better process control, and low sludge production (Gulamhussein et al., 2023).
1.10.2.5. Moving Bed Biofilm Reactor (MBBR)
MBBR technology integrates biofilm-attached growth mechanisms with a suspended carrier medium, thereby enhancing biomass retention and functional diversity while minimizing spatial requirements (di Biase et al., 2019). Empirical data and comprehensive reviews have consistently demonstrated that aerobic MBBR systems exhibit superior efficacy in the removal of a diverse array of pharmaceuticals, including several antiretrovirals (ARVs), when compared to conventional activated sludge processes. This is particularly evident when MBBRs are employed as a tertiary treatment stage following secondary treatment processes. Laboratory and pilot-scale studies focusing on ARVs have reported significant reductions in the concentrations of compounds such as tenofovir, nevirapine (62%), tenofovir (74%), efavirenz (94%), ritonavir (95%), and emtricitabine (95%) from South African wastewater within a 10-day MBBR polishing period (Mokgope et al., 2022). The high levels of degradation efficiency are ascribed to the adsorptive capacity of the biofilm matrix and the extended solids retention times facilitated by the carriers (Tang et al., 2017).
Integrating these technologies with existing conventional wastewater treatment facilities can achieve nearly complete mineralization of pharmaceutical compounds. The incorporation of AOPs would augment the current treatment processes, providing rapid degradation, minimal sludge production, and the capability to eliminate micropollutants that are resistant to traditional biological treatment methods. The modular and adaptable design of AOPs facilitates their incorporation as tertiary polishing steps within current wastewater frameworks, thereby substantially enhancing the quality of effluent without necessitating considerable reconstruction. Ideal for the South African context, where constraints on resources, population growth, and increased usage of ARV drugs pose challenges to wastewater treatment capacity. Hence, the authors advocate for AOP systems as a practical and economically feasible strategy to achieve nearly complete pharmaceutical removal while sustaining financial and operational viability.
1.10.3. Policy and Practice Recommendations
Beyond technological solutions, a coordinated effort is required to enhance ARV monitoring and management. ARV surveillance should be formally integrated into the National Toxicity Monitoring Programme, with findings linked to the Green Drop certification system to incentivize improved wastewater performance. Hospitals, clinics, and pharmaceutical companies should adopt sustainable disposal practices to minimize contamination, while public awareness campaigns should encourage responsible disposal of medications. Interdepartmental collaboration between the Department of Water and Sanitation (DWS), Department of Health (DoH), and Department of Environmental Affairs (DEA) is essential to align water quality governance with HIV treatment programmes and environmental protection. Together, these measures directly contribute to achieving the United Nations Sustainable Development Goals (SDGs 3, 6, and 14), ensuring that South Africa advances sustainable water management while safeguarding public health and social equity.
Further, based on the global scale of the HIV epidemic and the extensive use of ARVs, international cooperation is crucial. Collaborative research and unified environmental standards for pharmaceutical contaminants can significantly improve how we assess and address the environmental risks ARVs pose to aquatic ecosystems. South Africa currently lacks explicit regulatory thresholds for ARVs in wastewater or surface waters. While the National Environmental Management: Waste Act (Act No. 59 of 2008) broadly governs waste discharge, the DWS issues general water use licenses. These frameworks do not explicitly address pharmaceuticals like ARVs. The 2023 Green Drop Report from DWS (Department of Water and Sanitation, 2023) indicates a significant number of WWTPs are underperforming, which raises concerns about their ability to manage pharmaceutical loads effectively. For example, it was reported that 64% of wastewater treatment works (WWTWs) are at high or critical risk of discharging partially treated or untreated water into rivers and the environment. The number of WWTWs in the high- and critical-risk categories has increased since 2013. This has negative environmental implications and poses risks to human health, e.g., cholera outbreaks are normally associated with wastewater pollution of water resources (Department of Water and Sanitation, 2023).
Additionally, no South African guideline values exist for environmental concentrations of ARVs in aquatic systems. In contrast, international benchmarks (e.g., from the European Medicines Agency (EMA)) for pharmaceuticals in surface waters proposed a threshold value of 10 ng/L. However, South African studies have recorded ARV concentrations far exceeding this value (e.g., nevirapine at 166 ng/L and lopinavir at 42 ng/L (Nibamureke and Barnhoorn, 2025). The EMA guideline has two phases: Phase I decision tree concludes with the calculation of a Predicted Environmental Concentration in surface water (PECSW), based on the predicted use of the product. When this PECSW is ≥ the action limit of 10 ng/L, a Phase II assessment should be performed. If the PECSW value is < 10 ng/L and no other environmental concerns are apparent, it is assumed that the medicinal product is unlikely to represent a risk to the environment following its recommended usage in patients, and no further risk assessment is required (European Medicines Agency, 2024).
To date, no formal water-quality guidelines for pharmaceuticals or ARVs exist in South Africa or the Southern African Development Community (SADC) region. This regulatory gap hampers the ability of agencies to enforce standards for ARV levels in the environment. We, therefore, propose interim threshold values for high-priority ARVs based on international risk assessment trigger values at 10 ng/L. Until locally validated guidelines are developed, this value can serve as a precautionary benchmark for environmental monitoring and compliance.
Building on NWU’s recommendations (North West University, 2025) for developing ARV-specific guidelines, improving wastewater treatment technologies, and pursuing multidisciplinary research, this review proposes additional measures. These include integrating ARV monitoring into the National Toxicity Monitoring Programme (NTMP), incorporating ARV thresholds into the Green Drop framework, incentivizing utilities to adopt advanced treatment technologies such as ozonation and nanofiltration, and fostering interdepartmental collaboration between DWS, DoH, and DEA to align water quality governance with the United Nations Sustainable Development Goals (SDGs 3, 6, and 14). To strengthen regulatory oversight and mitigate ARV contamination, we propose the following steps for national regulatory agencies:
- Develop South Africa-specific environmental quality standards (EQS) for key ARVs (e.g., nevirapine, efavirenz, tenofovir) using international risk assessment trigger values (RATVs) as benchmarks and adjusting for local ecotoxicological data.
- Set maximum allowable concentration thresholds in effluents for high-risk ARVs.
- Mandate routine monitoring of pharmaceutical pollutants (including ARVs) at wastewater treatment facilities through the NTMP.
- Update permitting criteria under the National Water Act to include pharmaceutical contaminants as part of water use licensing conditions.
- Integrate ARV discharge data into the Green Drop certification process, requiring regular pharmaceutical audits for facilities to maintain good standing.
- Promote interdepartmental collaboration among DWS, DOH, and DEA to ensure holistic water quality governance.
Such measures would align public health and environmental goals while also positioning South Africa as a leader in addressing pharmaceutical contamination in the Global South.
CONCLUSION
The growing detection of antiretroviral drugs in South Africa’s aquatic environments signals an emerging environmental and public health challenge that parallels the country’s success in HIV/AIDS management. The persistence of ARVs such as efavirenz, nevirapine, and lamivudine in wastewater effluents and receiving waters underscores the inefficiency of conventional treatment processes and the lack of regulatory oversight for pharmaceutical micropollutants. Ecotoxicological data reveal species-specific and concentration-dependent effects on algae, invertebrates, and fish, with chronic exposures capable of impairing growth, reproduction, and biochemical functions even at environmentally relevant concentrations.
Addressing this issue requires a multi-pronged strategy: (1) comprehensive monitoring of ARVs and their transformation products across water matrices; (2) nationwide ecotoxicity testing to generate predicted no-effect concentrations for environmental risk assessments; (3) adoption of advanced treatment technologies such as photocatalytic and oxidative processes tailored for resource-limited wastewater treatment plants; and (4) the establishment of ARV-specific regulatory thresholds within South Africa’s water quality framework. Strengthening intersectoral collaboration between the health, water, and environmental sectors will be essential to mitigate ARV pollution without undermining HIV treatment coverage.
Ultimately, protecting South Africa’s water resources from ARV contamination is not only an environmental imperative but also a public health necessity. Integrating pollution control into the country’s HIV response will safeguard ecosystem resilience, sustain human health, and uphold the long-term sustainability goals of equitable healthcare and clean water access.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: N.M. and P.N.: Contributed equally to the conception, literature review, writing, and editing of this manuscript. Both authors have read and approved the final version of the manuscript for submission.
LIST OF ABBREVIATIONS
| 3TC | = Lamivudine |
| ABC | = Abacavir |
| ACV | = Acyclovir |
| AOPs | = Advanced oxidation processes |
| APIs | = Active pharmaceutical ingredients |
| ART | = Antiretroviral therapy |
| ARVs | = Antiretroviral drugs |
| ATV | = Atazanavir |
| COFA | = N-(4-carbamoyl-2-imino-5-oxoimidazolidin) formamido-n90 methoxy-acid |
| d4T | = Stavudine |
| ddI | = Didanosine |
| DEA | = Department of Environmental Affairs |
| DoH | = Department of Health |
| DTG | = Dolutegravir |
| DWS | = Department of Water and Sanitation |
| EC50 | = Effective Concentration 50% |
| EFV | = Efavirenz |
| EMA | = European Medicines Agency |
| EQS | = Environmental quality standards |
| FTC | = Emtricitabine |
| GC–MS | = Gas Chromatography–Mass Spectrometry |
| HIV | = Human immunodeficiency virus |
| IDV | = Indinavir |
| LC50 | = Lethal Concentration 50% |
| LC–MS | = Liquid Chromatography–Mass Spectrometry |
| LPV | = Lopinavir |
| MECs | = Measured environmental concentrations |
| NTMP | = National Toxicity Monitoring Programme |
| NVP | = Nevirapine |
| PECs | = Predicted environmental concentrations |
| PNECs | = Predicted no-effect concentrations |
| RAL | = Raltegravir |
| RATVs | = Risk assessment trigger values |
| RQ | = Risk quotient |
| RTV | = Ritonavir |
| SDGs | = United Nations Sustainable Development Goals |
| SQV | = Saquinavir |
| TAF | = Tenofovir alafenamide |
| TDF | = Tenofovir disoproxil fumarate |
| TPs | = transformation products |
| WWTPs | = Waste water treatment plants |
| ZDV or AZT | = Zidovudine |
CONFLICT OF INTEREST
Ntombikayise Mahaye is the Associate Editorial Board Member of The Open Environmental Research Journal.
ACKNOWLEDGEMENTS
Declared none.


